Anvisa recently approved the texts of the draft resolutions of the Board of Directors and Normative Instruction regarding the new standard of labeling for food and beverages in Brazil. The approval establishes several changes in the labeling standard and clarifies several definitions of the formulations and how to communicate the nutritional values on the packaging.
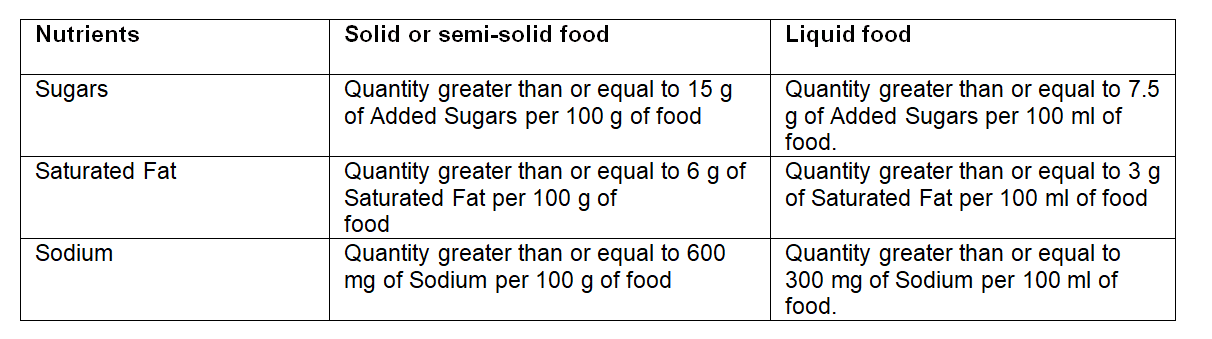
The main change is the new models for the nutrition label on the front of the packaging. The new label will become mandatory for foods and beverages whose amounts of Added Sugars, Saturated Fat, and Sodium are equal or higher than the limits defined in the Normative Instruction Draft. The table below shows these limits for food and beverage products.
LIMITS OF ADDED SUGARS, SATURATED FATS, AND SODIUM FOR NUTRITION LABEL ON THE FRONT OF THE PACKAGING – BRAZIL, OCTOBER 2020.
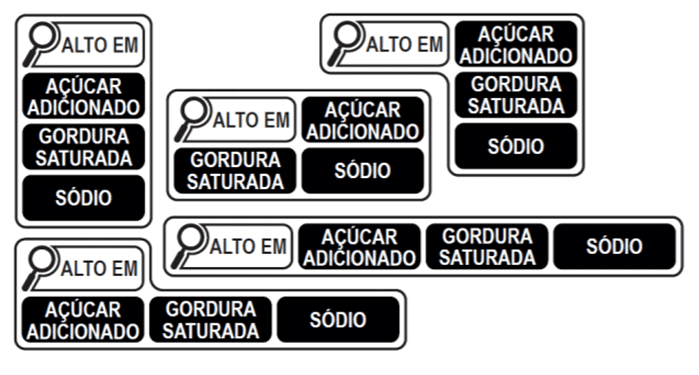
MODELS THAT SHOULD BE USED IN FOODS WHOSE AMOUNTS OF THE THREE NUTRIENTS (ADDED SUGARS, SATURATED FATS, AND SODIUM) ARE EQUAL TO OR GREATER THAN THE DEFINED LIMITS – BRAZIL, OCTOBER 2020.
As announced by Anvisa, the Nutrition Facts table will also undergo significant changes. The first of these is that the table will now have only black letters and white background to make it easier to read. Another significant change will be the information on the table. The identification of Total and Added Sugars, the number of Calories and Nutritional Value per 100 g or 100 ml (making it easier to compare with other products), and the number of portions per package will become mandatory.
The new labeling standard will highlight even more nutrients that were already being avoided by Brazilian consumers. According to Mintel research on healthy eating trends in Brazil, more than nine in 10 consumers showed interest in food and beverages with low sodium (with nearly half of them willing to pay more for such products) and low sugar content.
Regarding the deadline for adjusting to the new regulation, Anvisa will publish the new rules in the next days in the Diário Oficial da União, through a Resolution of the Collegiate Board of Directors (RDC) and a Normative Instruction (IN). The new rules will come into force 24 months after publication. When the new rules start, the products already on the market will have 12 months to adapt.
The regulatory agency also highlights that products intended exclusively for industrial processing or the foodservice should be adapted as soon as the new rules start. This requirement ensures that manufacturers can access the nutrition facts of raw materials and food ingredients used in their products. Products manufactured by small companies, such as family farmers and micro-entrepreneurs, have 24 months to adapt after the new rules start, totaling 48 months. For non-alcoholic beverages in returnable packaging, the adjustment may not exceed 36 months after the resolution comes into force.